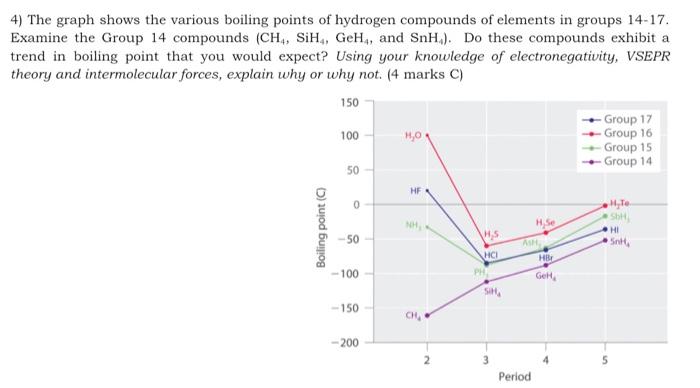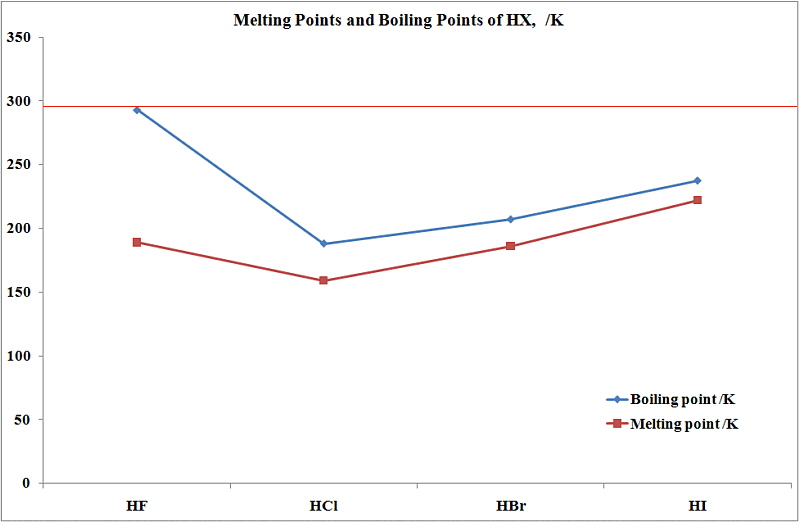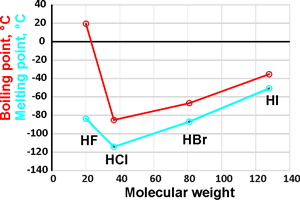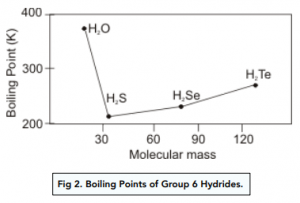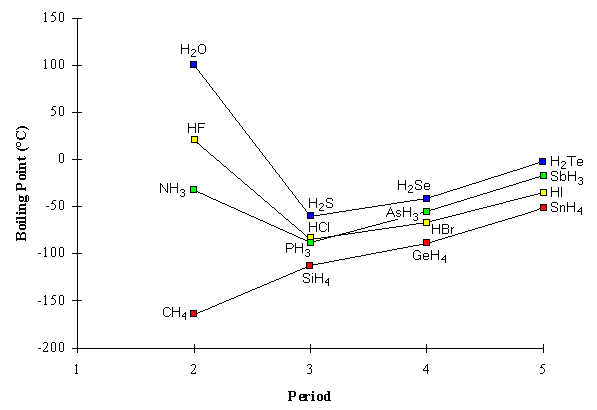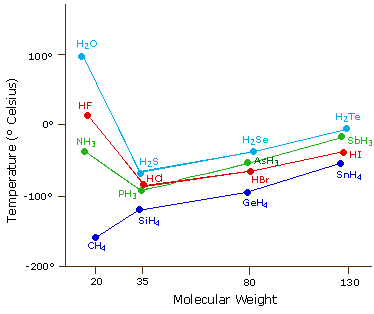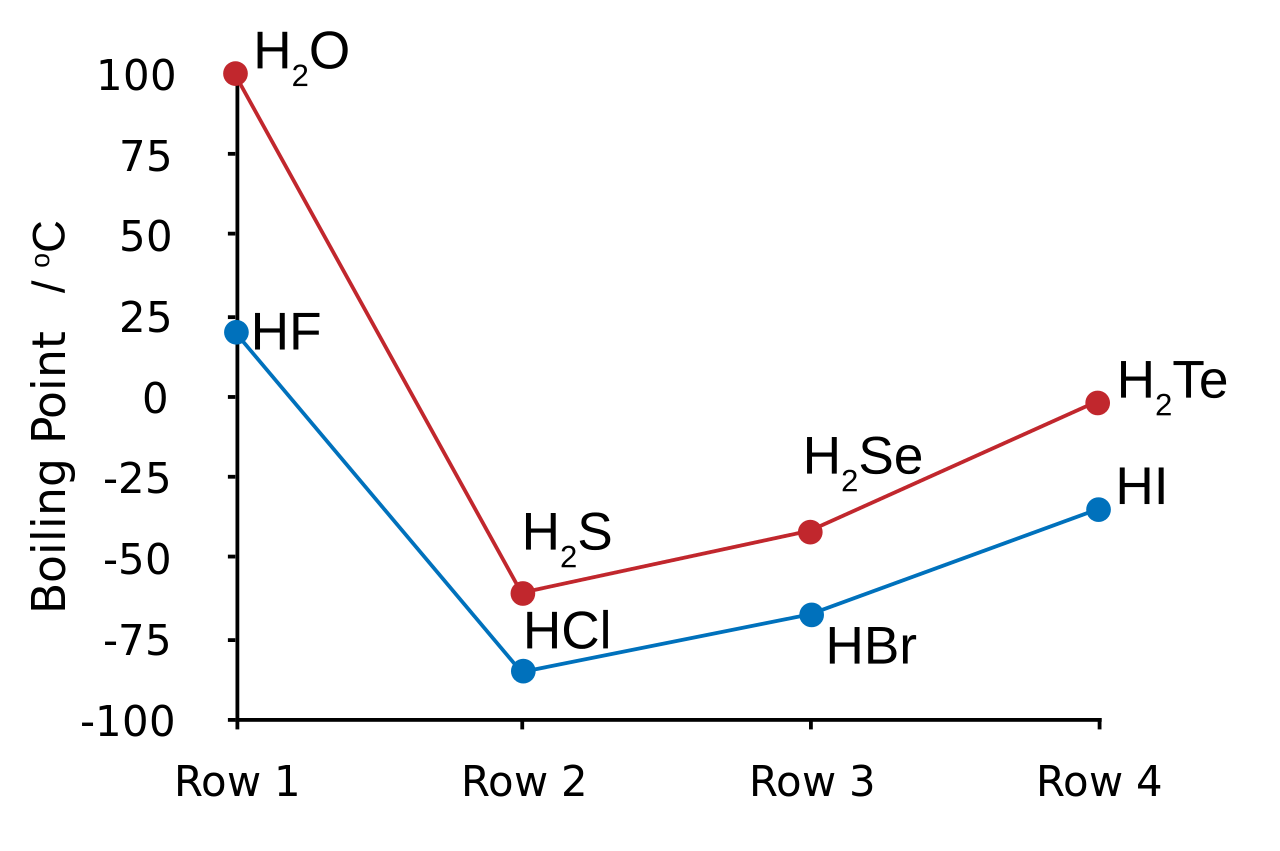
The liquefied hydrogen halides have the normal boiling points given above. The relatively high boiling point of HF can be correctly explained by which of the following? | Socratic
Boiling points of the hydrogen containing compounds of Group 14 (♢), 15... | Download Scientific Diagram
What will be the boiling point of water if hydrogen bonding between hydrogen and oxygen atoms is not present? - Quora

Hydrogen bonding, evidence from boiling points covalent hydride molecules water ice anomalous density hydrogen fluoride ammonia permanent dipole big difference in electronegativity propanone trichloromethane A level chemistry revision notes